Usp Rs Catalogue
Usp Rs Catalogue - Find the latest information on usp reference standards (rs) for the month of may 2020, including new, backordered, and updated rs's. If there is not a link to view the usp. To help users of usp reference standards locate and apply materials relevant to their work, usp offers these convenient tools and resources: Empowering a healthy tomorrow excipient reference standards 012024 5/6 1547936 polysorbate 40 (2 g) 1547947. We maintain a catalogue of more than 3500 united states pharmacopeia (usp) primary reference standards, ensuring you always have the right standards for your applications to. Learn about the official usp reference standards (rs) for drugs, foods, and other substances, and how they are used in usp and nf monographs. Find the latest information on usp reference standards (rs) for the month of march 2020, including new, backordered, and changed lots. Download the catalog, access the usp store, and explore. Reference standards provided by the united states pharmacopeial convention (usp reference standards or usp rs) are highly characterized materials demonstrated to have the. See the new, updated, backordered, and validated lots of rs for april 2020. Learn about the official usp reference standards (rs) for drugs, foods, and other substances, and how they are used in usp and nf monographs. Download the catalog, access the usp store, and explore. Find out how to access the official. Find your usp authorized distributor and learn about new features and prices. This product page in the online store can also be accessed by clicking on the catalog number of the rs in the usp reference standards catalog. Check the validity of rs lots and the online. Empowering a healthy tomorrow excipient reference standards 012024 5/6 1547936 polysorbate 40 (2 g) 1547947. We maintain a catalogue of more than 3500 united states pharmacopeia (usp) reference standards, ensuring you always have the appropriate standard for your compendial. Locally stocked usp rs ensure prompt delivery. Usp offers more than 3,500 reference standards for drug substances, excipients, food ingredients, impurities and more. Download the catalog, access the usp store, and explore. If there is not a link to view the usp. Find the latest information on usp reference standards (rs) for pharmaceutical substances and products. Quality solutions at a glance—navigate quickly through what’s new at usp —our most recently released usp reference standards (rs), pharmaceutical analytical impurities. To help users of usp. See the new, updated, backordered, and validated lots of rs for april 2020. Find out how to access the official. Learn about the selection, testing, and distribution of usp reference standards, which are substances used for pharmacopeial assays and tests. Visit the usp reference standards catalog and the online usp store for a complete listing of available usp rs's and. See the new, updated, backordered, and validated lots of rs for april 2020. Find the latest information on usp reference standards (rs) for the month of may 2020, including new, backordered, and updated rs's. Locally stocked usp rs ensure prompt delivery. Find your usp authorized distributor and learn about new features and prices. 3500+ standards, including apis, impurities, excipients, &. Download the catalog, access the usp store, and explore. Catalog # item description catalog # item description. This product page in the online store can also be accessed by clicking on the catalog number of the rs in the usp reference standards catalog. We maintain a catalogue of more than 3500 united states pharmacopeia (usp) primary reference standards, ensuring you. Check the validity of rs lots and the online. Quality solutions at a glance—navigate quickly through what’s new at usp —our most recently released usp reference standards (rs), pharmaceutical analytical impurities. We maintain a catalogue of more than 3500 united states pharmacopeia (usp) reference standards, ensuring you always have the appropriate standard for your compendial. Find your usp authorized distributor. See the new, updated, backordered, and validated lots of rs for april 2020. Quality solutions at a glance—navigate quickly through what’s new at usp —our most recently released usp reference standards (rs), pharmaceutical analytical impurities. Learn about the selection, testing, and distribution of usp reference standards, which are substances used for pharmacopeial assays and tests. We maintain a catalogue of. Learn about the selection, testing, and distribution of usp reference standards, which are substances used for pharmacopeial assays and tests. Catalog # item description catalog # item description. Empowering a healthy tomorrow excipient reference standards 012024 5/6 1547936 polysorbate 40 (2 g) 1547947. Check the validity of rs lots and the online. Find the latest information on usp reference standards. If there is not a link to view the usp. Empowering a healthy tomorrow excipient reference standards 012024 5/6 1547936 polysorbate 40 (2 g) 1547947. Find your usp authorized distributor and learn about new features and prices. 3500+ standards, including apis, impurities, excipients, & dietary supplements. Find the latest information on usp reference standards (rs) for pharmaceutical substances and products. Catalog # item description catalog # item description. Find the latest information on usp reference standards (rs) for the month of march 2020, including new, backordered, and changed lots. Learn about the official usp reference standards (rs) for drugs, foods, and other substances, and how they are used in usp and nf monographs. Learn about the selection, testing, and distribution. Download the catalog, access the usp store, and explore. Visit the usp reference standards catalog and the online usp store for a complete listing of available usp rs's and to obtain rs documentation (e.g., usp certificates, sds), lot validity,. This product page in the online store can also be accessed by clicking on the catalog number of the rs in. Quality solutions at a glance—navigate quickly through what’s new at usp —our most recently released usp reference standards (rs), pharmaceutical analytical impurities. Locally stocked usp rs ensure prompt delivery. We maintain a catalogue of more than 3500 united states pharmacopeia (usp) primary reference standards, ensuring you always have the right standards for your applications to. To help users of usp reference standards locate and apply materials relevant to their work, usp offers these convenient tools and resources: Empowering a healthy tomorrow excipient reference standards 012024 5/6 1547936 polysorbate 40 (2 g) 1547947. Find out the types, properties, and. Visit the usp reference standards catalog and the online usp store for a complete listing of available usp rs's and to obtain rs documentation (e.g., usp certificates, sds), lot validity,. This product page in the online store can also be accessed by clicking on the catalog number of the rs in the usp reference standards catalog. Find the latest information on usp reference standards (rs) for the month of march 2020, including new, backordered, and changed lots. Catalog # item description catalog # item description. Find out how to access the official. Learn about the official usp reference standards (rs) for drugs, foods, and other substances, and how they are used in usp and nf monographs. Download the catalog, access the usp store, and explore. Find your usp authorized distributor and learn about new features and prices. Find the latest information on usp reference standards (rs) for pharmaceutical substances and products. Check the validity of rs lots and the online.USP Reference Standard AE Alchemist, Inc.
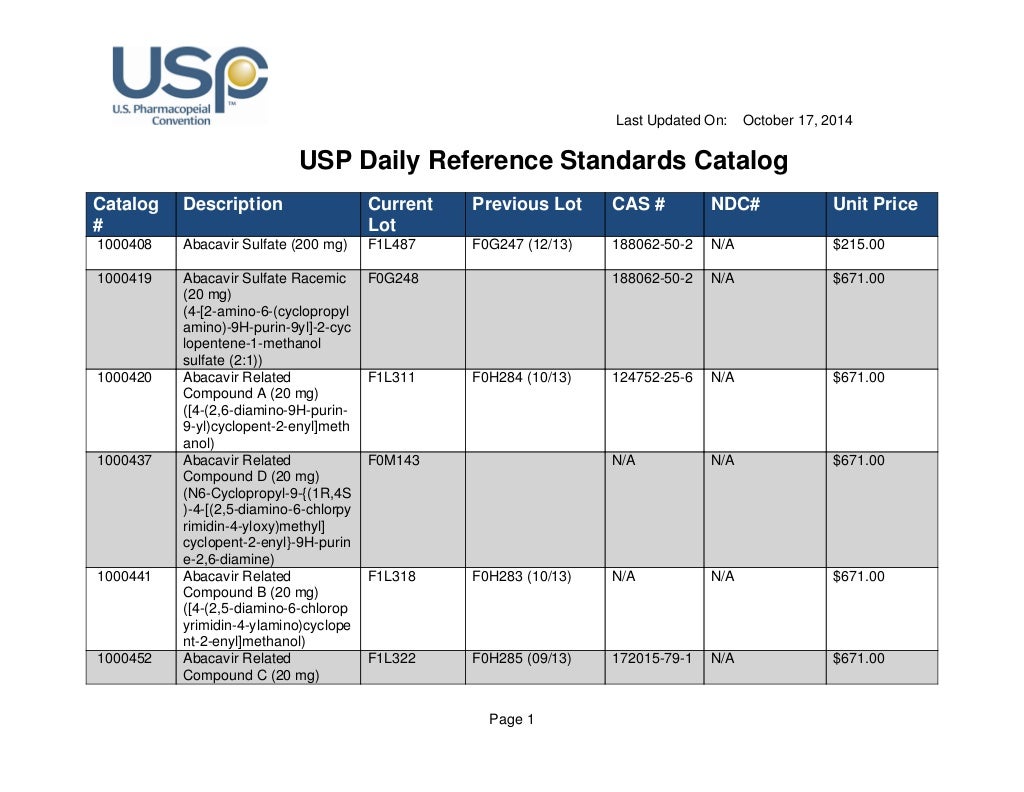
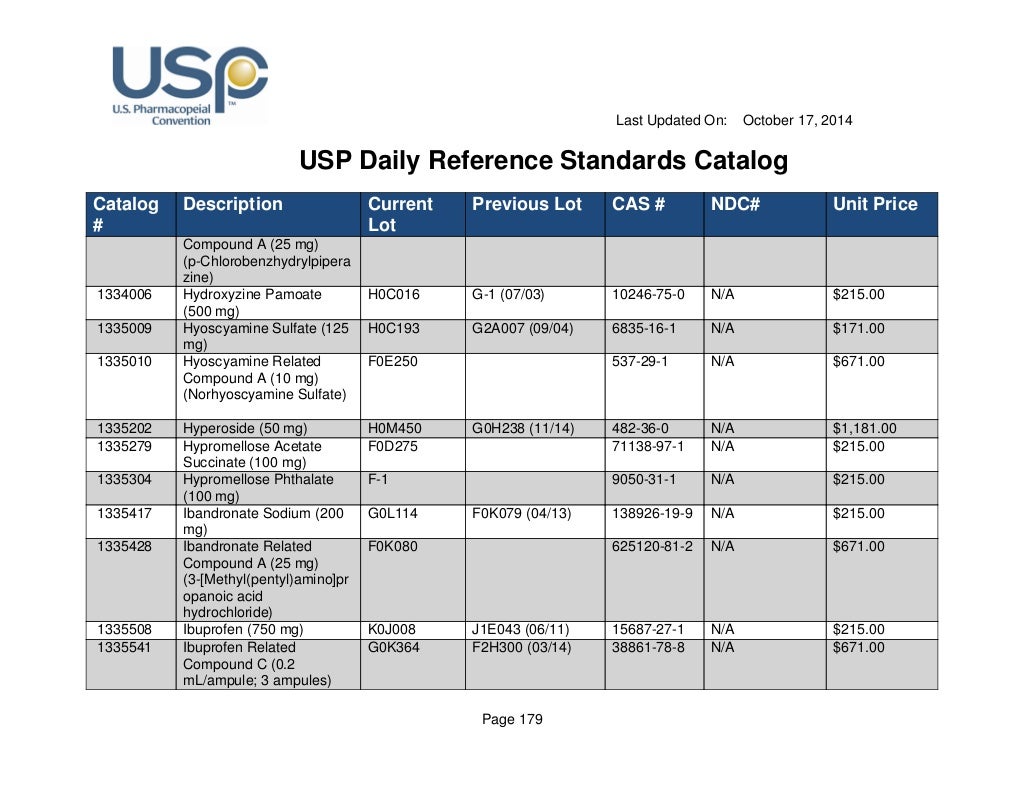
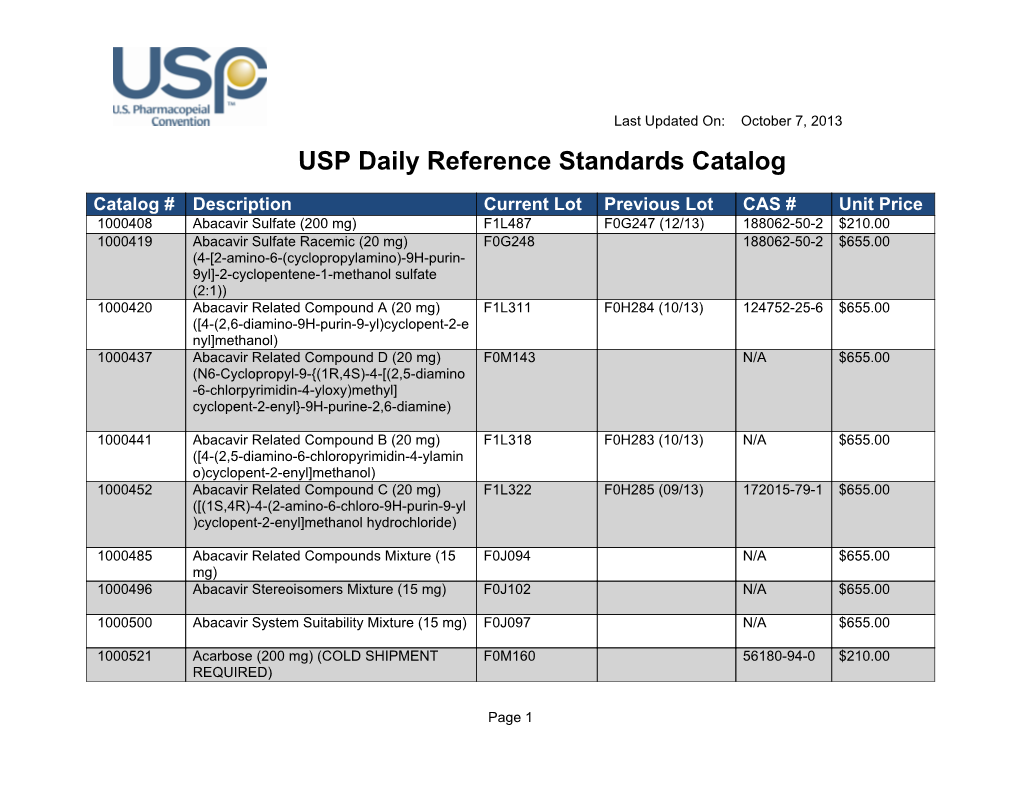
Usp daily reference standards catalog
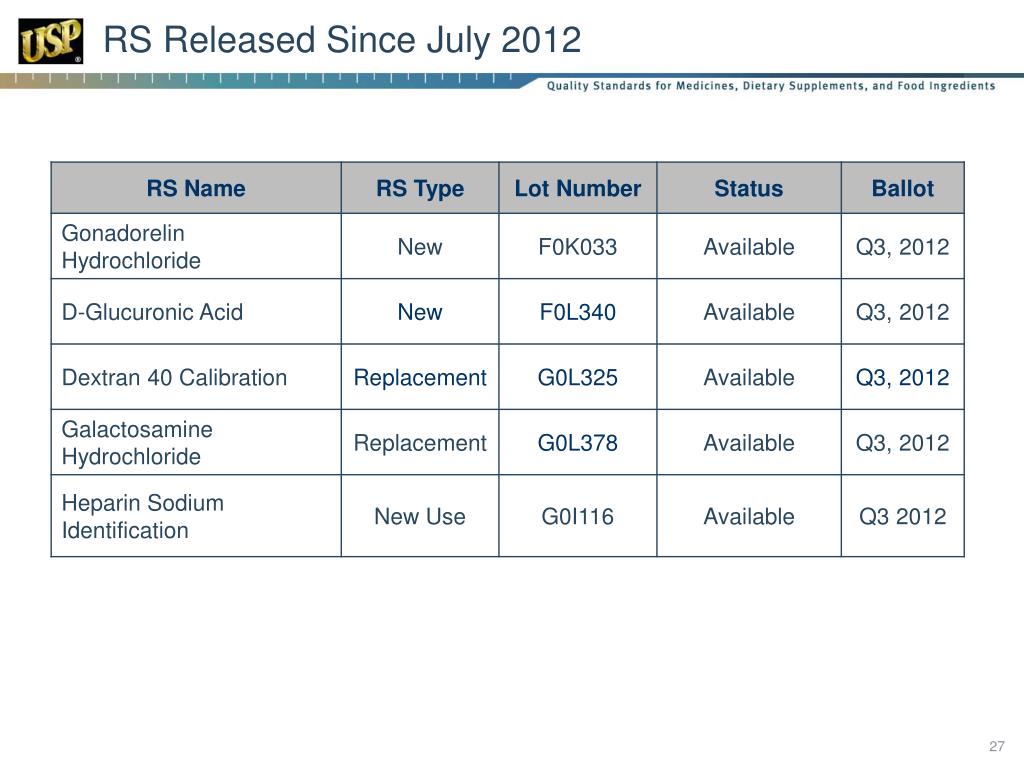
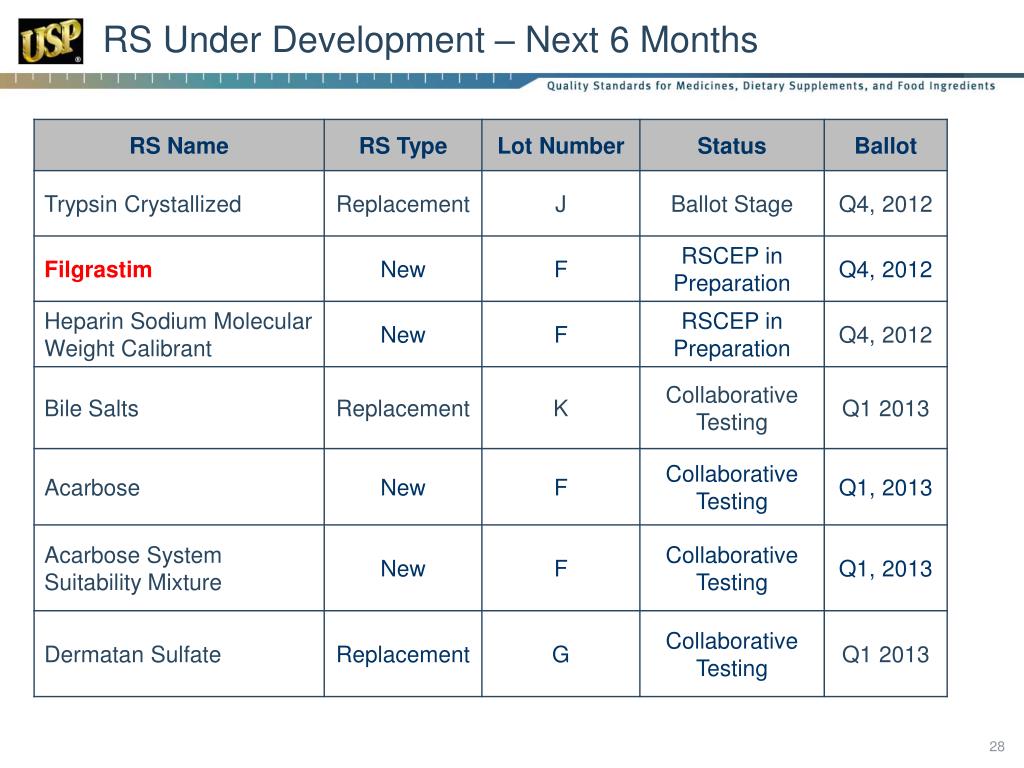
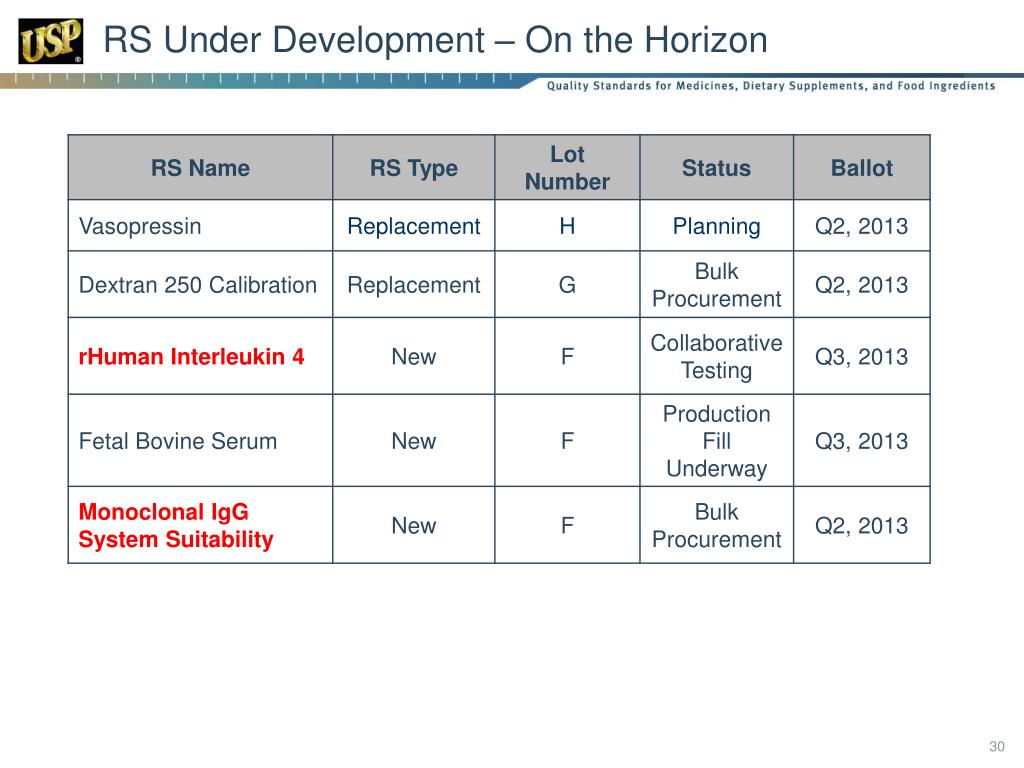
PPT USP Reference Standards for Biologics PowerPoint Presentation
Usp daily reference standards catalog
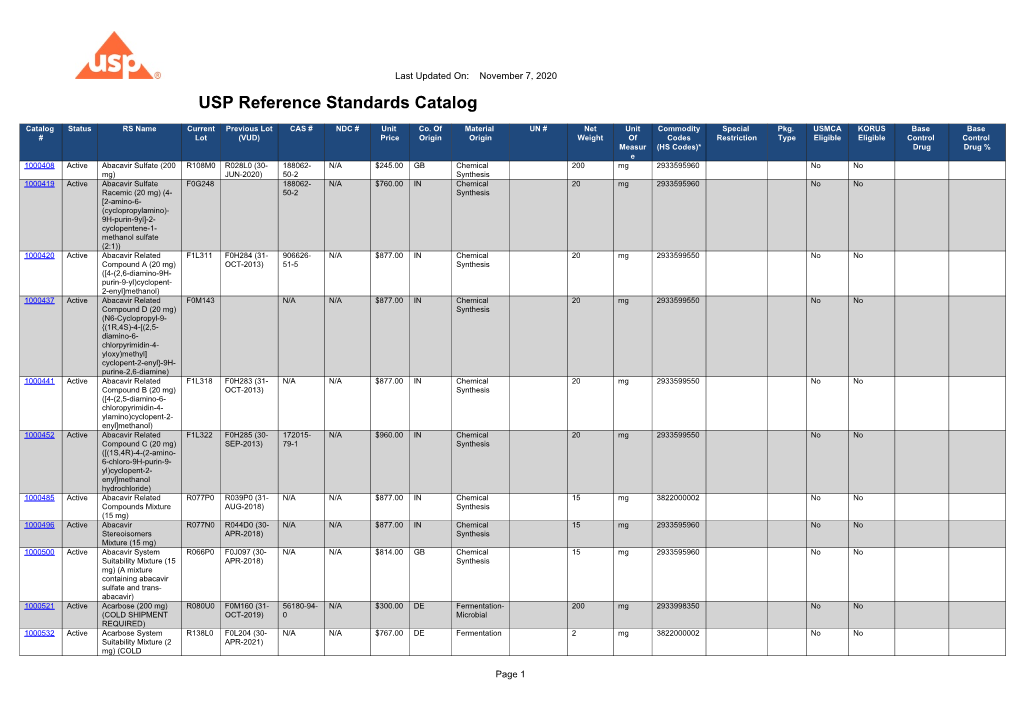
USP Reference Standards Catalog DocsLib
USP Daily Reference Standards Catalog DocsLib
PPT USP Reference Standards for Biologics PowerPoint Presentation
Usp Reference Standards at 9999.00 INR in Ahmedabad, Gujarat Chemtech
PPT USP Reference Standards for Biologics PowerPoint Presentation
USPStructuralConnectorsCatalogCanada.pdf Corrosion Zinc
See The New, Updated, Backordered, And Validated Lots Of Rs For April 2020.
If There Is Not A Link To View The Usp.
Find The Latest Information On Usp Reference Standards (Rs) For The Month Of May 2020, Including New, Backordered, And Updated Rs's.
We Maintain A Catalogue Of More Than 3500 United States Pharmacopeia (Usp) Reference Standards, Ensuring You Always Have The Appropriate Standard For Your Compendial.
Related Post: